In the pharmaceutical industry, speed is everything. The faster you can develop a new drug, test it in a clinical trial, and get it to market, the better the outcomes are for the patients and the business alike.
But that all hinges on the ability to access data. Without insights, none of this is possible – let alone at a meaningful speed or scale. If you’re manually writing, reviewing, and updating data policies as access requests pile up, you’re sacrificing speed for security. If you’re blowing through access requests as quickly as possible to minimize your to-do list, you’re compromising security for speed.
In pharma, where data compliance regulations and patient privacy are non-negotiable, you simply can’t afford to gamble on this tradeoff.
I know from working with major pharma companies that keeping up with data demands, let alone maintaining regulatory compliance, feels overwhelming. More than half of data professionals feel inundated with data policy management, and the issue is most acute in highly regulated industries. The rapid adoption of AI will only continue to drive up demand for data.
But as much as AI may create this challenge, it can also help solve it. In this blog, we’ll look at how AI-enabled data provisioning alleviates the burden of access management – so you can get back to mining insights for the next breakthrough in patient care.
Data analytics in the pharmaceutical industry is broken
Like many regulated enterprises, including financial services firms and public sector agencies, pharmaceutical companies face unique governance challenges. They are trusted to handle sensitive clinical trial data, complex genomic datasets, and protected health information (PHI) while complying with evolving regulatory frameworks such as HIPAA, GxP, and FDA guidelines. And, given the nature of the industry, they need to act quickly in order to deliver positive patient and public health outcomes.
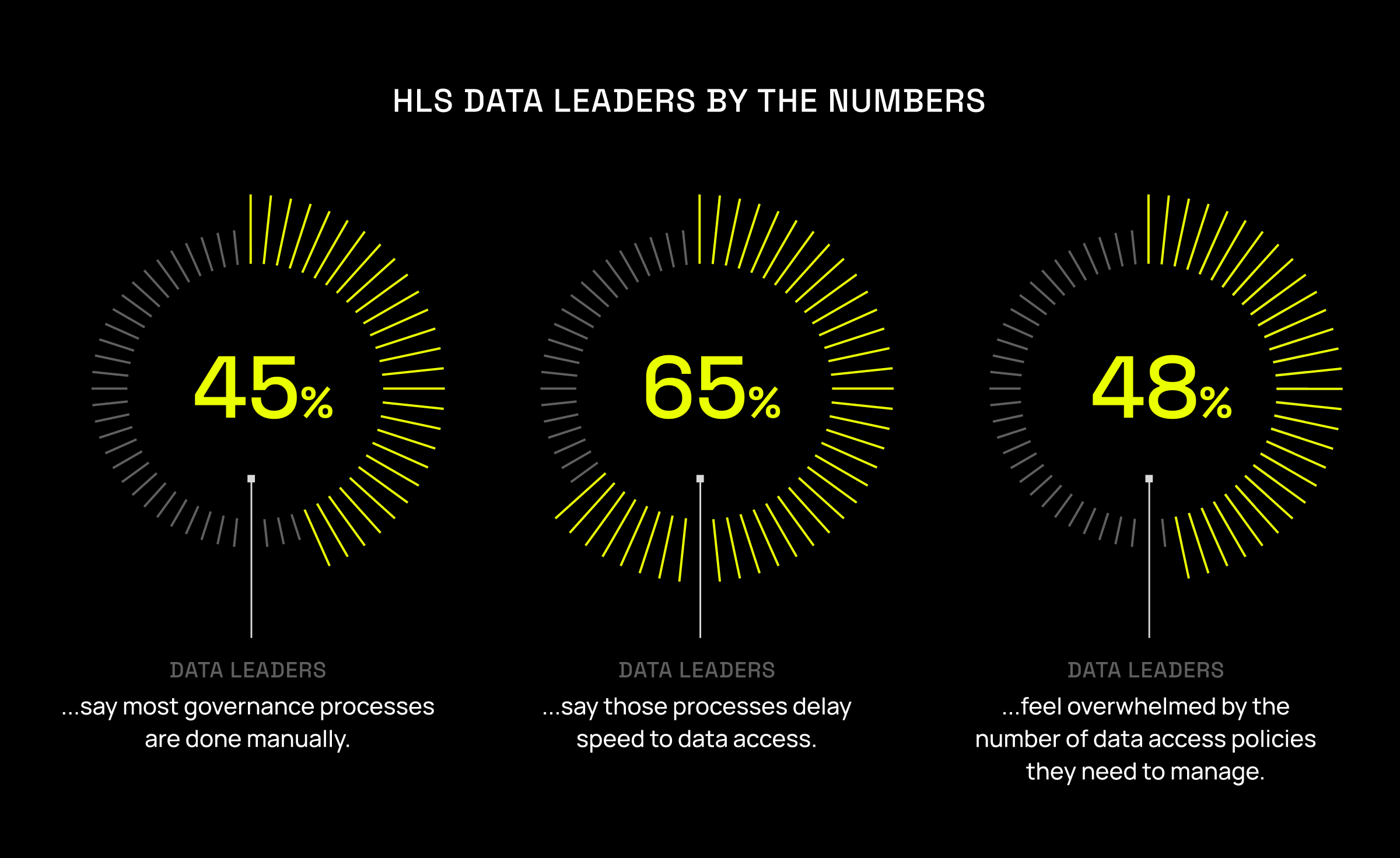
The problem is that many pharma companies are stuck in the manual era. In the healthcare and life sciences industry:
- 45% of data professionals say most of their governance processes are done manually.
- 65% say those very processes delay users’ data access.
Whether these organizations rely on platform-native processes, IT ticketing systems, or another ad hoc workflow, their approaches create bottlenecks, inconsistent policy enforcement, and compliance risks. This can result in significant downstream impacts, such as:
- Clinical trial delays due to inefficient data provisioning
- Late drug approval submissions stemming from compliance challenges
- Reputational damage and regulatory scrutiny following a privacy breach
As more humans become data consumers and AI agents autonomously request data, these consequences will have an outsized impact on what organizations are able – or unable – to do with their data. Traditional tooling, static policies, and manual processes simply weren’t built to handle the exponential growth in data demand that’s on the horizon.
The 4 challenges of pharmaceutical data management
As new technologies and AI applications emerge to improve data analytics in the pharmaceutical industry, they are also exposing the gaps in existing systems. Four rise the top that are critical for pharma, but relevant for any regulated industry:
- Data is hard to find
- Risk classifications are inconsistent
- Data volumes are massive
- Auditing is a non-negotiable headache
The good news is that while AI is contributing to the complexity, it can also be a solution. Let’s look at how.
Solving pharmaceutical data provisioning challenges with AI
When we think about using AI to resolve data provisioning challenges, it’s important to not conflate it with automation. Instead, think of AI as a multiplier – a tool that can bridge data discovery, provisioning, and auditing so that pharma data workflows run seamlessly, without additional overhead or human intervention.

1. Data is hard to find → AI-powered sensitive data discovery
Pharma data is rarely contained within a single location. It can live in trial management systems, clinical databases, genomic repositories, and external partner environments, to name a few. The problem is that trying to locate what you need, when you need it, can be a time-consuming headache. Even with a comprehensive data catalog, the gap between discovery and access remains a blocker (that’s where a data provisioning platform comes in).
AI-driven data discovery scales your ability to quickly scan connected datasets, tag sensitive attributes, and dynamically enforce policies based on metadata. This takes the onus off data product owners, governors, and stewards to manually comb through datasets, and reduces the risk of sensitive information falling through the cracks.
2. Inconsistent risk classifications → AI-informed risk classification frameworks
Building a data classification framework allows you to maintain oversight of diverse and highly sensitive pharmaceutical datasets. But if the risk levels laid out within the framework are inconsistently applied, the framework won’t help you much.
Manual classification processes are subjective and often lack context, increasing the likelihood of sensitive data exposure, inadequate protection, and non-compliance.
However, AI models trained on pharma-specific taxonomies, regulatory requirements, and historical tags, are able to consistently apply risk classifications, flag anomalies for human review, and continuously learn from new data patterns. That means fewer false positives, fewer oversights, and fewer concerns about provisioning sensitive data.
3. Data volumes are massive → AI-assisted data provisioning
Nearly half of data professionals in the healthcare industry report feeling overwhelmed by the number of data access control policies they need to manage. And with every clinical trial or research initiative generating terabytes of new data, the strain will continue to grow. Manual processes and ticketing systems – which were meant for app access, not data access – are bound to break.
AI systems that assist reviewers by separating high- or medium-risk access requests from low-risk ones alleviate the burden while accelerating access. These systems leverage data and user metadata, as well as free-text request justifications, to generate a recommendation about whether to approve, temporarily approve, or deny access. Reviewers can then bulk approve low-risk requests, and prioritize the ones that require more scrutiny – saving time for them and the requestors.
AI copilots can also author policies based on plain-text prompts, using tags and metadata to dynamically enforce the right policies – right when they’re needed. In pharmaceutical settings, this immediacy can be the difference between getting a new drug to market in time, or missing the window when it can have the highest impact.
4. Auditing is a non-negotiable headache → AI-driven monitoring and auditing
In the pharmaceutical industry, auditing and compliance are table stakes. Two-thirds of healthcare-based data professionals report being subject to 10+ data regulations, and 70% cite strengthening regulatory compliance as a high priority. Still, monitoring data access, generating audit reports, and proving compliance can be a massive, time-consuming headache.
One of the most important – and most dreaded – compliance tasks is access recertifications. That’s because they require periodically combing through who has access to what data in order to determine whether specific users or groups still warrant access permissions. When done manually – which it often is – it can take hours, days, or even weeks to complete (again, on a regular basis), while other tasks and new access requests fall by the wayside.
With AI, the process becomes more seamless and less painful. AI systems automatically:
- Aggregate user and data metadata
- Assess the frequency of user requests
- Determine whether those requests align with intended data usage purposes
- Make a recommendation on whether to recertify access or revoke it
AI-driven access recertifications, along with always-on monitoring, unified audit logs, and flags for anomalous behavior, help make auditing and compliance less of a burden – without giving up control entirely.
The future of AI-driven data provisioning in the pharma industry
Breakthroughs in the pharmaceutical industry have always been driven by data – how quickly and safely you can get the right data into the right hands. AI will just make those data transactions and innovative breakthroughs happen even faster – if it’s controlled and put to work the right way.
What does this look like in practice?
- Clinical trial data is continuously collected, aggregated, and analyzed by AI to quickly identify trends and provide recommendations on whether or how to shift directions.
- Updates to FDA or EMA guidance are immediately recognized by AI and reflected in data access policies, without the need to manually rewrite policies or retag data.
- AI agents use data on public health and drug inventory levels to trigger vaccine production and supply chain coordination so that medicines get to market before a virus outbreak.
- AI assistants pull data on who accessed what data, when, and why for auditors, providing natural-language explanations and flags for potential compliance concerns.
Most importantly, though, the AI-driven future of data provisioning in the pharmaceutical industry is one in which access reviewers and approvers are able to do their jobs without being consumed by them.
Access requests don’t pile up.
Reviews aren’t rushed or inconsistent.
Sensitive data exposure isn’t left to chance.
Governance and provisioning are seamless, automatic, and immediate.
Pharma companies that focus on AI-driven data provisioning workflows now will move faster, deliver better patient outcomes, and outpace competitors because they’ll be able to wrangle and make sense of their data – responsibly, compliantly, and at scale.
The time to get ahead of AI-fueled data demands by harnessing and implementing AI systems for pharma workloads is now. And who knows? It could lead to the next lifesaving breakthrough in pharmaceutical research and development.
Learn more.
Get the latest on AI-driven data provisioning solutions.






